The same groups at the University of Oxford and the Jenner Institute that developed the AstraZeneca-Oxford Covid-19 vaccine (Covishield) reported that their investigational malaria vaccine, R21/Matrix-M, or R21/MM, was 77% effective in children age 5 and 17 months in a 12-month Phase 2 clinical trial in Burkina Faso1. Previously, it was shown to be safe and produce an appropriate immune response in Phase 1 clinical trial2. Both vaccines use a genetically engineered adenovirus and the Matrix-M adjuvant. Billions of doses are being manufactured by the Serum Institute of India (SII) and will be sold at a low price. The goals of this article are to describe the R21/MM vaccine and how malaria is very different than Covid-19.
Malaria is very different than Covid-19
Covid-19 has dominated the news due to its mortal threat to the elderly. Children under 17 are least likely to die from Covid-19, while people who are over 85 years old have died at a much higher rate that is 8700 times that of children in the USA. At the same time, the Americas have suffered the most deaths per capita by Covid-19, while Africans have the least – partly because of their younger population.
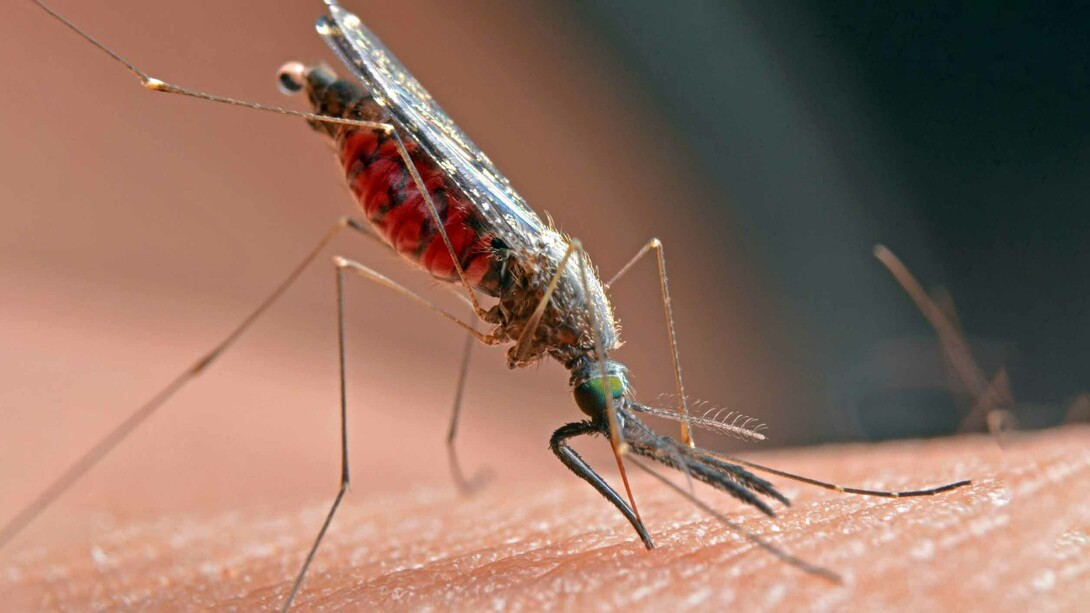
Malaria is caused by parasitic protozoans in the genus Plasmodium. Protozoan is an informal term for organisms that consist of a single eukaryotic cell that does not contain filaments (like molds). They are more complex than prokaryotes (bacteria and archaea). Eukaryotic cells have internal organelles, including a cell nucleus, unlike prokaryotes. Both prokaryotes and eukaryotes are living cells, containing DNA and RNA. Moreover, they are self-making, or autopoietic3. Plasmodium protozoans are carried by infected female mosquitoes of the genus Anopheles. The infected mosquito transmits a sporozoite (the infective form of the Plasmodium protozoan) to the human host. Sporozoites travel through the blood and reach the liver. They reproduce asexually, producing thousands of merocytes, which infect new red blood cells. This initiates a series of asexual multiplication cycles that produce more new, infective merocytes. Some merocytes develop into immature precursors of male and female reproductive cells, or gametes. When a fertilized mosquito bites an infected person, gametocytes are taken up with the blood and mature in the mosquito’s gut. The male and female gametocytes fuse and form a fertilized zygote, made from the fertilization event by two gametes. The zygotes develop into new sporozoites that migrate to the mosquito’s salivary glands, ready to infect a new host. Viruses like the SARS-CoV-2 do nothing like this. They contain either DNA or RNA, but not both. They are not capable of autopoiesis. They are completely dependent on cells within their host to make themselves and replicate.
There are five parasite species of protozoans that cause malaria in humans. Two of them cause the most damage. In 2018, Plasmodium falciparum (P. falciparum) accounted for 99.7% of malaria cases in the WHO African Region, 50% of cases in the WHO South-East Asia Region, 71% of cases in the Eastern Mediterranean and 65% in the Western Pacific. Plasmodium vivax is the predominant parasite in the Americas, causing 75% of malaria cases there. In 2019, there were an estimated 229 million cases and 409,000 deaths caused by malaria, worldwide4. In contrast to Covid-19, children under the age of five are the most vulnerable. According to the WHO, 94% of malaria cases and deaths were in Africa.
Malaria is preventable and curable. However, if not treated within 24 hours, it can progress to severe illness, often leading to death. It has been much more difficult to develop a safe and effective vaccine against malaria than Covid-19. Our adaptive immune system produces memory T cells that help provide long-term immunity by completely eliminating or killing the virus5. Unlike the HIV virus that causes AIDS, the SARS-CoV-2 virus does not remain in the body. Similarly, the human body can’t completely eliminate the protozoan P. falciparum. Once a person is infected, some of this protozoan remains, even if he or she is cured of the infection in red blood cells, you can still be re-infected6. In the case of malaria, successful T cell memory does not develop. Scientists are still trying to understand why.
Currently, there is only one malaria vaccine that has been approved by the European Medicines Agency (EMA)7. It’s called the RTS,S vaccine, RTS,S/AS01, and Mosquirix. It contains a specific fusion protein called RTS,S. It contains a portion of a protein made by circumsporozoites. It is a secreted protein of the sporozoite stage. It is fused with hepatitis B surface antigen (RTS), and combined with hepatitis B surface antigen (S) to make the entire RTS,S protein. Also, about 80% of the vaccine contains hepatitis surface antigen protein monomers that are not fused to any other protein. Mosquirix targets the pre-erythrocyte stage of malaria. It targets malaria parasites after they enter the human body after being released from the salivary glands of a feeding mosquito. This is before the pathogenic blood-stage of the infection. However, the WHO does not recommend the routine use of this vaccine in babies between 6 and 12 weeks of age8. Therefore, RTS,S/AS01 has not yet been prequalified for use by WHO, but instead a Malaria Vaccine Implementation Programme (MVIP) was launched in Malawi, Ghana and Kenya over the course of 20191.
The R21/MM vaccine
There are many children living in a malaria-endemic area with high transmission in Burkina Faso1. So, it was appropriate to perform a controlled, double-blind phase 2 clinical trial on the R21/MM vaccine there. Like Mosquirix, it is a pre-erythocyte vaccine that targets the pre-erythrocyte stage of malaria. R21/MM and RTS,S both include hepatitis B surface antigen fused to the secreted protein of the sporozoite stage. However, R21/MM does not contain excess hemoglobin fusion protein monomers. It also contains the same Matrix-MTM saponin-based adjuvant that is in the Novavax Covid-19 vaccine9. It stimulates both humoral and cellular immune responses to vaccines. It stimulates the entry of antigen-presenting cells into the injection site and enhances antigen presentation in local lymph nodes, thus boosting immune response. Novavax plans to deliver 10 million doses of NVX CoV2373 that could be used in Phase 2/3 trials, or under an Emergency Use Authorization if approved by the FDA. They can increase that to 100 million if needed. They have a recent history of success - a quadrivalent influenza (flu) vaccine.
In phase 2 clinical trial in Burkina Faso, 450 children were recruited into 3 groups; Group 1 received 5µg R21 and 25µg MM, Group 2 received 5µg R21 and 50µg MM and the control group, Group 3, received rabies vaccinations. Doses were administered from early May to early August 2019, largely prior to the seasonal peak of malaria transmission starting in July. Safety, immunogenicity, and vaccine efficacy is being assessed over 24 months with the primary efficacy endpoint after 6 months following the primary series of vaccinations (3 doses). All participants also received a booster vaccination approximately 12 months after their third vaccination, prior to the start of the following malaria season. In the higher-dose adjuvant group, the efficacy was 77%. In the lower dose adjuvant group, it was 71%. The vaccine had no serious adverse side effects1.
Development of a malaria vaccine based on RNA technology
Various types of RNA technology can be used to develop new vaccines, such as the Moderna and Pfizer/BioNTech vaccines for Covid-1910. They used messenger RNA (mRNA) that encodes the spike protein produced by the SARS-CoV-2 virus. The spike protein activates a person’s immune system, as it develops a memory for this antigen. The P. falciparum protozoan produces very different proteins than the virus. One of the most intriguing has a very similar structure to that of the macrophage migration inhibitory factor (MIF), a cytokine found in humans11. It’s puzzling that a simple protozoan would have a specialized protein that is important in the immune system of humans. Plasmodium species produce an ortholog of the cytokine macrophage migration inhibitory factor, PMIF, which modulates the host inflammatory response to malaria12. Orthologs are genes in different species that evolved from a common ancestral gene.
Scientists studied a strain of malaria that was produced in the laboratory which lacked PMIF11. In experimental models, the strain-infected mice had some disease, but a memory T cell response did occur. This showed that the function of this PMIF protein is to kill memory T cells. So, they prepared a vaccine candidate based on self-replicating, self-amplifying RNA or saRNA. It’s an mRNA that encodes PMIF and replication proteins. By combining the saRNA with the PMIF antigen, they were able to demonstrate full protection in mouse models. They are now collaborating with the Oxford Vaccine Group to evaluate it11.
There is a possible advantage of malaria vaccines. They probably offer a much better option than killing all the mosquitoes on Earth. That is, scientists are developing ways to do this using CRISPR and other technologies13. Mosquitoes have always been the deadliest animal to humans. Mosquitoes in the Anopheles genus cause not only malaria, but also dengue and chikungunya. Other mosquitoes cause yellow fever, Zika, West Nile and encephalitis. Together, these mosquitoes have killed more people than any other animal on Earth – including humans with all our wars. Still, eliminating several entire species could have unpredictably bad effects on the ecosystems in which mosquitoes live.
In conclusion, even though Covid-19 continues to dominate the news, scientists and doctors continue to develop vaccines against other deadly diseases, such as malaria. However, it’s much more difficult to develop vaccines against malaria caused by a living cell than Covid-19 which is caused by the comparatively simple and new parasite, the SARS-CoV-2 virus. Mosquitoes, Plasmodium species and malaria have existed much longer.
Notes
1 Datoo, M.S. et al. High efficacy of a low dose candidate malaria vaccine, R21 in 1 Adjuvant Matrix-MTM, with seasonal administration to children in Burkina Faso. The Lancet, 21 April, 2021.
2 Venkatraman, N. et al. Phase I assessment of first-in-human administration of a novel malaria anti-sporozoite 2 vaccine candidate, R21 in matrix-M adjuvant, in UK and Burkinabe volunteers. MedRxiv, 2019.
3 Luisi, P.L. The quest for wholeness. The systems view and the search for spirituality. Wall Street International, 23 May, 2017.
4 World Health Organization. Malaria. 1 April, 2021.
5 Smith, R.E. Covid-19: Immune response to the SARS-CoV-2 virus. The response to this virus depends in large part on the health of one’s neuroendocrine immune system. Wall Street International, 24 April, 2021.
6 Mahase, E. A vaccine against malaria: five minutes with Richard Bucala. BMJ, formerly British Medical Journal, Volume 372, Article n. 651, 8 March, 2021.
7 European Medicines Agency, Mosquirix H-W-2300, 2015.
8 World Health Organization. Malaria vaccine: WHO position paper, January 2016.
9 Smith, R.E. Developing vaccines and treatments for Covid-19. Progress report. China starts vaccinating military personnel. Wall Street International, July, 2021.
10 Smith, R.E. Vaccines based on modern RNA technology. This technology's potential for vaccines and other diseases. Wall Street International, 24 December, 2020.
11 Mahase, E. A vaccine against malaria: five minutes with Richard Bucala. BMJ, formerly British Medical Journal, Volume 372, Article n651, 8 March, 2021.
12 Garcia, A.B. et al. Neutralization of the Plasmodium-encoded MIF ortholog confers protective immunity against malaria infection. Nature Communications, Volume 9, Article 2714, 2018.
13 Smith, R.E. Using CRISPR gene editing to create new foods. An important part of the fourth industrial revolution. Wall Street International, 24 May, 2019.